41 2 step reaction energy diagram
Label the energy diagram for a two-step reaction - Learn ... Mar 19, 2018 — Label the energy diagram for a two-step reaction. ... An energy profile diagram is a theoretical representation that shows how the energy of the ... 5.6: Reaction Energy Diagrams and Transition States May 30, 2020 — The energy diagram for a typical one-step reaction might look like this: ... “transition state,” as the two are often used interchangeably.
Phase (matter) - Wikipedia Distinct phases may be described as different states of matter such as gas, liquid, solid, plasma or Bose–Einstein condensate.Useful mesophases between solid and liquid form other states of matter.. Distinct phases may also exist within a given state of matter. As shown in the diagram for iron alloys, several phases exist for both the solid and liquid states.
2 step reaction energy diagram
Potential Energy Diagrams | Chemistry for Non-Majors | | Course Hero Figure 1. A potential energy diagram shows the total potential energy of a reacting system as the reaction proceeds. (A) In an endothermic reaction, the energy of the products is greater than the energy of the reactants and ΔH is positive. (B) In an exothermic reaction, the energy of the products is lower than the energy of the reactants and ... Potential energy diagram for a two-step sequential reaction Potential energy diagram for a two-step sequential reaction A 1 19 3 50 AB* 4 33 3 40 B 3 28 3 30 BC* 4 41 3 20 C 5 13 3 10 3 0 the red lines are error bars on the x -value Adjust energies Derived from Scott Sinex's Excel chart 1 2 3 4 5 0 10 20 30 40 50 Energy Reaction coordinate step 1 step 2 A B C A B C k 1k 2 Question #62891 | Socratic The figure above represents the reaction profile of a two step, exothermic reaction. The y-axis represents the potential energy of the reaction species, and the x-axis represents the progress of the reaction. The reaction is exothermic because the energies of the products are lower than those of the reactants. The reactants are represented by the horizontal line at the far left of the graph ...
2 step reaction energy diagram. 1.2. Process Flow Diagram (PFD) | Diagrams for Understanding … 03/07/2012 · 1.2. Process Flow Diagram (PFD) The process flow diagram (PFD) represents a quantum step up from the BFD in terms of the amount of information that it contains. The PFD contains the bulk of the chemical engineering data necessary for the design of a chemical process. For all of the diagrams discussed in this chapter, there are no universally accepted … Arrhenius Theory and Reaction Coordinates - Chemistry 302 The key is that there are many many potential paths between reactants and products. The reaction coordinate represents the lowest energy path. For example, in the reaction of CH 3 Cl + OH- to form CH 3 OH and Cl-, the mechanism of this reaction is a single step in which the CH 3 Cl collides with the OH- and forms the products. We can envision a ... Energy Diagrams of Reactions | Fiveable Nov 23, 2021 · To find the activation energy, you should be looking for two numbers: the potential energy of the reactants and the energy of the activated complex (the maximum point). (energy of activation complex) - (PEreactants) (100 kJ) - (40 kJ) = 60 kJ In other words, it takes 60 kJ of energy to complete the reaction. Energy Diagram - Organic Chemistry Video | Clutch Prep Video explaining Energy Diagram for Organic Chemistry. ... Free energy diagrams show overall changes in potential energy during reactions. ... Determined by Gibbs Free Energy (ΔG ° = ΔH ° - TΔS). Kinetics describes rate. Determined by the activation energy of the rate determining step (E a). Example #1: Describe the favorability and rate ...
7.2 SN2 Reaction Mechanism, Energy Diagram and Stereochemistry The energy changes for the above reaction can be represented in the energy diagram shown in Fig. 7.1. S N 2 is a single-step reaction, so the diagram has only one curve. The products CH 3 OH and Br - are in lower energy than the reactants CH 3 Br and OH -, indicates that the overall reaction is exothermic and the products are more stable. Multistep reaction energy profiles (video) - Khan Academy Next we're ready for the second hill or the second step of our mechanism. In the second step, AC the intermediate AC reacts with D to form A and CD. So the top of this second hill would be the transition state for this second step. So we can show the bond between A and C braking, and at the same time the bond between C and D is forming. Solved Choose the energy diagram for a two-step reaction, A - Chegg Expert Answer 100% (36 ratings) Transcribed image text: Choose the energy diagram for a two-step reaction, A → B → C, in which the relative energy of the compounds is A< C < B, and the step A→ B is rate-determining. Select the single best answer. Reaction coordinate Reaction coordinate IV. SOLVED:Sketch an energy diagram for a two-step reaction in which both ... Problem 13 Medium Difficulty Sketch an energy diagram for a two-step reaction in which both steps are exergonic and in which the second step has a higher-energy transition state than the first. Label the parts of the diagram corresponding to reactant, product, intermediate, overall Δ G †, and overall Δ G ∘. Answer View Answer Discussion
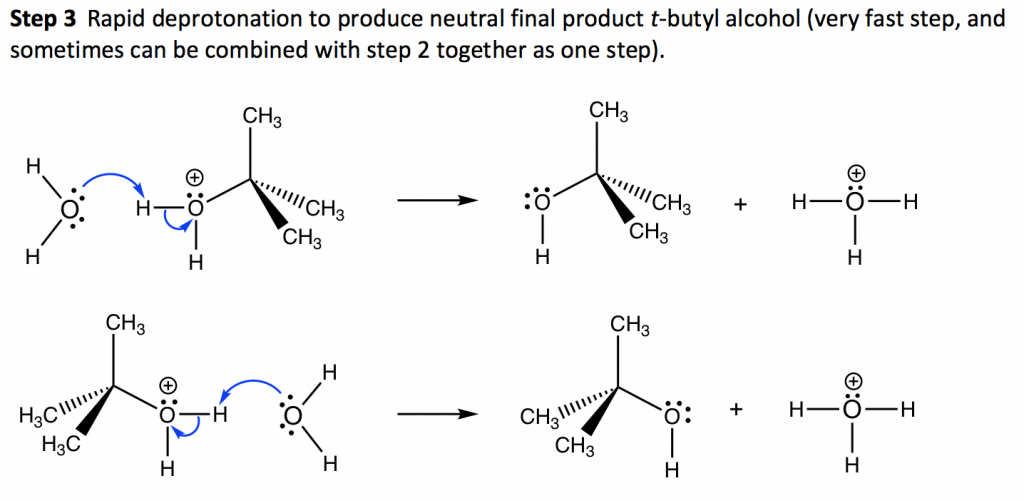
7.4 SN1 Reaction Mechanism, Energy Diagram and Stereochemistry 7.4 SN1 Reaction Mechanism, Energy Diagram and Stereochemistry SN1 Reaction Mechanism The reaction between tert -butylbromide and water proceeds via the SN1 mechanism. Unlike S N 2 that is a single-step reaction, S N 1 reaction involves multiple steps. Reaction: (CH 3) 3 CBr + H 2 O → (CH 3) 3 COH + HBr 6.8: Energy Diagram for a Two-Step Reaction Mechanism Jun 2, 2019 — A potential energy diagram for an SN1 reaction shows that the carbocation intermediate can be visualized as a kind of valley in the path of the ... Mechanisms and Potential Energy Diagrams | Chemistry for Non-Majors ... A potential energy diagram for a two-step reaction is shown and labeled. Practice View the section on two-step reactions at the site below and then do the self-test (both buttons are at the top of the slide). Don't worry about - just consider it an indication of activation energy as is in the diagram above. 18.15: Mechanisms and Potential Energy Diagrams Feb 21, 2022 — The reaction whose potential energy diagram is shown in the figure is a two-step reaction. The activation energy for each step is labeled ...
Multistep Reactions - Softschools.com The energy diagram of a two-step reaction is shown below. In the above reaction, a reactant goes through one elementary step with a lower activation energy (transition state 1) to form the intermediate. The intermediate then goes through a second step (transition state 2) with the highest energy barrier to form the product.
6.7: Reaction Coordinate Diagram for a Two-Step Reaction ... Jun 2, 2019 — A potential energy diagram for an SN1 reaction shows that the carbocation intermediate can be visualized as a kind of valley in the path of the ...
Energy Diagrams of Two Step Reactions - YouTube Watch Complete videos @ Organic Chemistry 1
Energy Diagram for a Two-Step Reaction Mechanism Jun 01, 2022 · Complete Energy Diagram for Two-Step Reaction A Two-Step Reaction Mechanism The transition states are located at energy maxima. The reactive intermediate B+ is located at an energy minimum. Each step has its own delta H and activation energy. The overall energy difference between the starting materials and products is delta H overall.
How do you find the rate determining step from a graph? The rate determining step in a reaction mechanism is the slowest step. It is characterized by its high activation energy. Consider the energy diagram represented below of a two-step mechanism. The first step is the slow step since it has the highest activation energy. Here is more about this topic in the following video: Chemical Kinetics ...
E1 Reaction Mechanism and E1 Practice Problems - Chemistry Steps Step 1: Loss of he leaving group. The energy diagram of the E1 mechanism demonstrates the loss of the leaving group as the slow step with the higher activation energy barrier: The dotted lines in the transition state indicate a partially broken C-Br bond. The Br being the more electronegative element is partially negatively charged and the ...
Label The Energy Diagram (7 Bins) And Indicate Which Reaction ... So, if the graph is higher in a given region.Energy Diagram for a Two-Step Reaction Mechanism In many reactions more than one step is involved in the formation of products. A Two-Step Reaction Mechanism We draw an energy diagram for each step, and then combine them in an energy diagram for the overall two step mechanism. ...
Draw an energy diagram for a two-step reaction, $$ A \righ | Quizlet Find step-by-step Chemistry solutions and your answer to the following textbook question: Draw an energy diagram for a two-step reaction, $$ A \rightarrow B \rightarrow C, $$ where the relative energy of these compounds is C < A < B, and the conversion of $$ B \rightarrow C $$ is rate-determining..
Energy Diagrams: Describing Chemical Reactions Draw an energy diagram for a two-step reaction that is exothermic overall, and consists of a fast but endothermic first step, and a slow but exothermic second step. Indicate DGrxn, as well as DG1* and DG2* for the first and second activation energies, respectively. Label the positions corresponding to the transition states with an asterisk.



Post a Comment for "41 2 step reaction energy diagram"